Case Studies: understanding & Strategy
First Time Contact to Patients from the Industry's View
Project brief
Nancy Meyerson-Hess is an internationally renowned expert for clinical operations and an Associated Partner of admedicum® Business for Patients. She has cooperated directly with patients related to clinical trial design, recruitment and retention on numerous occasions.
Situation
A company plans to develop a proprietary compound for a chronic disease. For the company, it is a new therapeutic area. Planning for the clinical development program has just started. To date no one from the R&D team has had direct contacts with patients.
Challenge
Finding the right way to get into contact with patients and to encourage them to share their stories with people they don’t know.
Objective
The clinical project team wants to learn from patients about their daily life with the disease and their experience with available treatment options. They hope to learn more about the medical need the company needs to address.
Procedure
Approach
A team delegate:
- Reaches out to key opinion leaders (KOLs). In this case, the KOLs can’t point them to patients who would be open to speak about their disease to a group of unknown people.
- Calls a patient organization. They are open to contact in general but suggest the company to send a written request with detail on what, where and how a meeting should take place and who should join. They will then consider to support such a meeting and check who would be best to join.
- Talks to a patient at a conference attended by patients and patient organizations. This patient agrees to tell her patient journey to a larger group of the company.
Evaluation
What went well
Going through a number of channels (KOLs, patient organization, conference) is very appropriate. Usually involved physicians and patient organizations are very helpful in making an introduction. The direct and personal contact to a patient in this case immediately opened the door to further interaction.
What can be improved?
Although the team was very credible, transparent and engaged in seeking patient insights, they had no real action plan: their approach was not embedded into a greater plan of why to do that, what to achieve with it and how to do it.
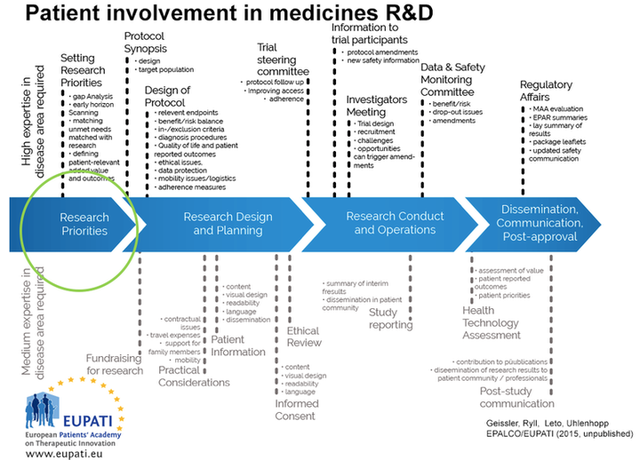
Modified source: EUPATI (2016): Guidance for patient involvement in ethical review of clinical trials; page 4
Considerations and recommendations
Preparing for a collaboration
Before you start engaging with patients, make a plan for the why, what and how. For first interactions, it doesn’t need to be very detailed. However, everybody in and outside your organization should be able to understand it. Be very clear on what you want and can commit to, and what not, and communicate this transparently.
Several sources have created checklists that can be used in preparing collaborations. Two good examples are:
- Checklist “Preparing a Collaboration” EUPATI
- Checklist “Preparing a Collaboration” ABPI and National Voices
A master checklist based on the EUPATI and ABPI & National Voices can be found below.
Before meeting patients or even reaching out, make sure you do your homework. A considerable part of the patient experience can be found online, in communities and other places. It is important that company employees pre-read these, so as not to disappoint patients when they come and find out that the companies employees do not even know the most basic elements of the patient experience. Pre-reading also helps to formulate questions, which allow to probe deeper (and consequently understand more) than "just" open listening.
Social media screening is a powerful tool to better understand who you need to talk to and what you need to learn more about. Go to the checklist "Mapping the Patient Landscape” for broader context.
Master checklist for establishing first time contact with patients
- Identify patients to be involved in the interaction: Tool “Patient identification” EUPATI.
- Ask your medical advisors and KOLs for their contacts to patient organizations and patient experts, internationally, and arrange personal introductions.
- Visit conferences with patient participation. A great conference to start with is Patients as Partners US or Europe. It is the only conference that exclusively focuses on patient-industry interaction in R&D.

- Search patient organizations’ websites for events and ask whether you can join (important: always make yourself transparent as a Biopharma company).
Start with some personal conversations before moving on to group events.
Please realize that patients may have concerns you need to address before they feel safe enough to speak up. Also, always be aware that not every patient may be equally capable as a patient representative. There are several lines along which patients can contribute and exert their influence: Tool "Defining Patient’s Role” DIA.
Partnering With Patients in the Development and Lifecycle of Medicines, THERAPEUTIC INNOVATION & REGULATORY SCIENCE, 2015 Nov; 49(6): 929–939
Defining patient centricity with patients for patients and caregivers: a collaborative endeavor, BMJ Innovations, March 24, 2017
Help us improve this guide
Submit Feedback
Tool
Considerations for Implementing Expert Patient/Patient Group Input
Recommended Contributors :
- Program leaders
- Patient liaisons
- Sponsor representatives
- Clinical investigators
- Research team
- Trial site staff
- IRB
- Expert patient(s)/Patient Group representatives
Communicating with Patients throughout the Program
- How does the phase of drug/biologic/device development process covered by this program impact communication with patients?
- What translation and/or cultural adaptions?
- Wat language will be used to communicate with and about the patients?
- Are research questions and procedures culturally sensitive and appropriate?
- How will patients be referred to (e.g. “subject” vs. “patient” vs. “participant”)?
- What is the communication plan for patients throughout the program?
- Message content
- Audience
- Messenger
- Delivery mechanisms
- Timing
- Feedback mechanisms
- What feedback mechanisms and processes are in place for the patients to comment on sites, investigators, and the study participant experience?
- What role will social media play in the communications?
- How is social media defined?
- What restrictions should there be, if any?
- How can social media be used to advantage (e.g. for trial recruitment, to educate patients)?
- What limits should be placed on use of social media, if any? Why?
- How will those limits be communicated and enforced?
- What methods will be used to interact with patients and other stakeholders?
- Focus groups
- Interviews
- Surveys
- Inclusion on advisory councils
- Inclusion in meetings with researchers
- What data/information can and will be shared with the patients and when?
- Aggregate (de-identified)
- Patient-specific
- What are the restrictions (propieratry and regulatory) constraining the release of data?
- How do we ensure that this information is shared in patient-friendly language? How will that be determined/monitored?
Additional resources
Communication Handbook for Clinical Trials.
Guidance for Biomedical HIV Prevention Trials, p 37-38: “Stakeholder education plan.”
Checklist
Preparing a collaboration
Defining the interaction
Patients, patient representatives and industry should take responsibility to ensure interactions are meaningful by clearly defined processes and actions, progressed to timelines. In addition, all participants should be prepared for the interaction.
Prior to each interaction, agree mutually on (where applicable):
- The objective of project involving patients and/or areas of common interest to establish agreed structured interaction, providing all parties with necessary protection with regards to independence, privacy, confidentiality and expectations (see section 11. written agreement)
- The type of input and mandate of the involved person
- The tools and methods of interaction, e.g. types and frequency of meetings, ground rules, conflict resolution, evaluation
- Desired patient / patient partner organisation to foster long-term working partnerships, with independence ensured (in scope)
- The profile of the type of patient/s or patient representative/s to be involved and their number
- How activity outputs will be used and ownership of outputs
- How and when the patient/s involved will be informed of outcomes
- Contractual terms and conditions including consent and compensation (see section 11, written agreement).
- Other elements according to the specific project
Source
Download
Checklist
Preparing a collaboration
The four key principles for collaboration:
1. Clarity of Purpose
Each party should be clear about the reason for and the planned outcome of the collaboration – and the ultimate benefit for patients
2. Integrity
Each party should act and be seen to act honestly and with integrity at all times
3. Independence
Each party should maintain their independence
4. Transparency
Each party should be open and honest about the purpose of the collaboration and be able to account publicly for the associated activities and any exchanges of funding
Using this guide: a checklist
- Has there been a frank discussion about the purpose and expected benefits of the collaboration, and any risks, addressing all the issues in this guide?
- Are the objectives and planned outcomes of the collaboration specified?
- Are the roles of each partner and reporting mechanisms specified?
- Has a written agreement or contract been put in place, which sets out how each party will adhere to the four key principles?
- Is there a named senior individual accountable for managing and maintaining the relationship and monitoring adherence to the four key principles?
- Is information about the collaboration published on the company and charity websites?
- Can each party confidently explain the collaboration in public?
Source: National Voices, The Association of the British Pharmaceutical Industry (ABPI) (2015): Working
Download
Checklist
Patient identification
There are many ways to identify patients to be involved in an interaction. The main routes are through:
- Existing patient organizations
- EUPATI or similar project
- Advertising opportunities for patient participation
- Medical Key Opinion Leaders. healthcare providers, hospitals and researchers and other institutions
- Systematic social media search for patient exchange platforms
- Unsolicited requests previously made by interested parties
- Existing advisory boards / groups (e.g. EFPIA Think Tank, Patients and Consumers Working Party at the EMA)
- Specialized patient engagement agencies
Source: European Patients’Academy on Therapeutic Innovation (EUPATI) (2016): Guidance for patient involvement for industry-led medicines R&D. (12/06/17)
Download
Tool
List of Useful Conferences
Important international conferences, attended by multi-stakeholder groups, including patient advocates
ECRD: European Conference on Rare Diseases and Orphan Products
Rare diseases conferences with an industry focus, but attended by patient advocates as well
The Global Orphan Drug Conference and Expo
You may want to attend conferences from patient groups to learn about their needs from their interactions and subjects. Many of these conferences are open for industry participants but one should keep in mind that these events are from patients for patients and industry should respect the guest status.
Example for an indication-specific conference by and for patients (industry welcome)
European Rett Syndrome Congress (international)
Also, many patient organizations welcome industry at their charity events as a sponsor and as participants, like at charity runs. Check out your area of engagement.
Tool
Defining the Patient’s Role
Although you may not have selected the expert patients or patient groups (EP/PGs) yet, outlining their roles and responsibilities at this stage helps to define your needs. Keep in mind that EP/PG roles may vary at different stages of the program or may evolve in response to new requirements. Once selected, discuss the roles with your EP/PGs to clarify what they can contribute based on their unique expertise and experience and avoid misunderstandings at the outset, e.g. if they’re expecting to have a partnership role but you’ve designed reactor role (see Types of Patient Roles chart below).
| Patient role | Examples | Engagement level |
| Partnership role |
| High |
| Advisor role |
| Moderate |
| Reactor role |
| Low |
| Trial or study participant |
| None |
Source: DIA (2017): Considerations Guide to Implementing Patient-Centric Initiatives in Health Care Product
Development. (02/06/17)
Download
Get in Touch
Contact admedicum
Checklist
Mapping the Patient Landscape
Following a general understanding of the patient journey and unmet needs and before systematically interacting with patients and KOLs in a specific therapeutic area a company should have an understanding of the stakeholder field. The checklist below can help you obtain such an understanding, by:

Source: This checklist has been developed by admedicum® Business for Patients GmbH.
Download
